Viscoelastic Properties of Vitreous
The vitreous humour is a viscoelastic gel-like fluid that fills the inner space of the eye and plays an important role in the development of many age-related ocular diseases. Its unique properties arise from the underlying macromolecular structure, a lightly cross-linked network of collagen and hyaluronan. Using rheology, changes in the material behavior are correlated to changes in the macromolecular structure to investigate underlying mechanisms in the development of vitreous pathologies and effects of drug treatments.
Collaborators: Jean Pierre Hubschman, MD (Jules Stein Eye Institute, UCLA)

Wetting Dynamics of Ultra-hydrophobic Surfaces and Spreading of Emulsions on Solid Substrates
Controlling the surface wettability of the ultra-hydrophobic surfaces has extensive industrial applications ranging from coating, painting and printing technology and efficiency increase in power and water plants. Controlling the surface wettability of ultra-hydrophobic surfaces requires enhancing the knowledge about the dynamics of wetting on these surfaces. We currently are doing experimental investigation on the dynamics of wetting on ultra-hydrophobic surfaces by looking deeply in to the dependency of the dynamic contact angles both advancing and receding on the velocity of the three-phase boundary (Solid/Liquid/Gas interface) using the Wilhelmy plate method applied in the tensiometer with impressing the motion on the three-phase boundary by moving the hydrophobic glass slide with different velocities. In our experiments we also focus on the effect of viscous forces on the system which could play an important role on the behavior of the dependency of advancing and receding contact angle on the velocities of the plate and for each constant velocity the variation of both advancing and receding contact angles with respect to depth of the immersed plate are investigated.
Surprisingly, there has not been any comprehensive study of emulsion spreading to date; this is due to the complexity of the structure of the emulsions and non-homogeneity of the dispersed phase bubbles in size as well as distribution through the emulsion. The spreading of water/silicone oil emulsions on glass substrates was investigated. The emulsions were prepared with varying volume fractions of water dispersed in silicone oil, with addition of small amounts of surfactant to stabilize the emulsion structure. The time dependent variation of dynamic contact angle, base diameter, and the spreading rate of the droplets of an emulsion are different from a pure substance. The effect of water/silicone oil weight percentage as well as the droplet size and dispersed phase bubble size were also investigated. The weight percentage of water/silicone oil emulsion and droplet size did not have significant influence on the spreading dynamics; however the dispersed phase drop size affected the spreading dynamics substantially.



The Spreading and Arrest of Molten Liquid on Cold Solid Substrates
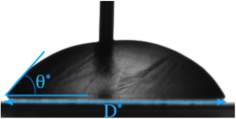
The physics of non-isothermal spreading followed by phase change, unlike that for isothermal spreading, isstill a mystery. We focus on the dynamic and thermal characteristics of liquid drop’s spreading and their subsequent arrest due to freezing. Spreading of liquid is recorded and the evolution of liquid spread diameter and liquid-solid contact angle are measured from the recordings of the Drop Shape Analyzer (DSA100) machine (Figure 1). After the initiation of solidification at the basal plane, the liquid drops pinned to the substrate showing fixed footprints and contact angles. A physical hypothesis using scaling analysis is provided to explain the relationship between the arrested base diameter and solidification contact angle (Figure 2) with respect to Stefan number. The Stefan number is described as the ratio of sensible to latent heat in a phase change process. The experimental observations of solidified drops on a cold substrates corroborate the derived physical theory.

Thermal Properties Measurement of Supercritical Fluids
This effort will result in empirical measurement of thermal properties of supercritical fluids within a well-defined, simplified experiment. These detailed measurements will supply benchmark data to the scientific community, and establish promising candidates for use in a supercritical energy storage system. In order to design the components of a thermal system operating at supercritical conditions, the behavior of supercritical fluids needs to be well understood. The objective is to find the thermal properties of various fluids in their supercritical state, which will be determine through high pressure and high temperature laboratory testing and analytical analysis. Both the thermal conductivity and thermal diffusivity of fluids will be obtained by using the hotwire method. This effort will provide useful information for the design of the system components, as well as for the selection of a viable fluid candidate with suitable properties and heat transfer characteristics applicable to high temperature, high pressure thermal energy storage.
Funding: ARPA-e
Collaborators: Richard Wirz and Adrienne Lavine (UCLA), Gani Ganapathi (JPL)

Copyright (c) Pirouz Kavehpour 2015