Motivations
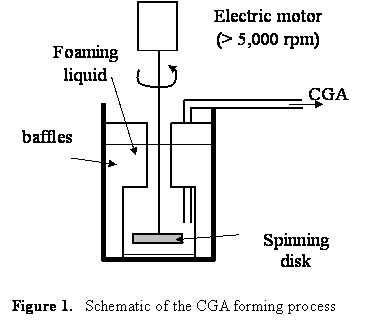
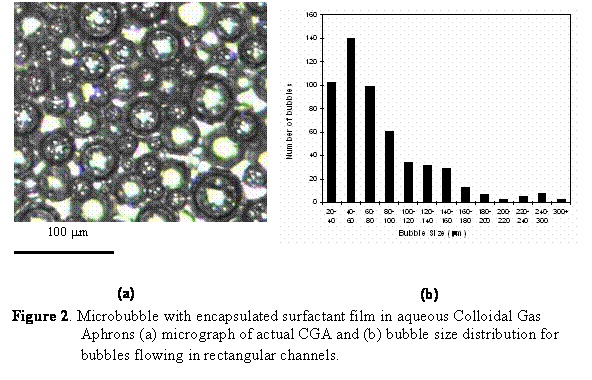
Colloidal Gas Aphrons (CGA), also called microfoams, consist of bubbles between 10 and 100 mm in diameter with a porosity of up to 70%. They can be produced by stirring an aqueous surfactant solution contained in a fully baffled beaker at room temperature, as described in Figure 1. The solution is stirred by a spinning disk driven at 5,000 to 10,000 rpm by an electrical motor. Two to four baffles are equally spaced around the container and extend well above the surface of the solution. Once a critical stirring speed has been reached, waves are produced at the liquid surface. The waves beat up against the baffles and are forced to re-enter the liquid at the baffles. It is believed that the re-entering liquid carries a thin film of gas which becomes sandwiched between the liquid and the baffle. Such a thin film is unstable and breaks into microscopic bubbles encapsulated by a soapy shell, i.e. minute gas aphrons. After a few minutes of stirring, CGA forms and rises at the surface of the solution. It has been reported that CGAs made of water and surfactant solutions (i) have stable bubbles with a narrow and reproducible size distribution between 10 and 100 mm (see Figure 2), (ii) exhibit high stability, (iii) separate easily from the bulk liquid phase, and (iv) can be easily pumped through tubes while keeping their structure. However, even though the flow properties of CGA have been discussed qualitatively in the literature, to the best of our knowledge, no quantitative data on the rheology, pressure drop, and friction factor associated with CGA flow in pipes has been reported.
Most applications of colloidal gas aphrons take advantage of (i) their large interfacial area, (ii) the adsorption of particles at the microbubble interfaces, and (iii) their stability for enhanced mass transfer. Possible applications include (1) separation, (2) oil recovery, (3) firefighting, (4) fermentation and bioreactors, (5) bioremediation, (6) material synthesis, and in general wherever macroscopic foams are currently being used.
Results
Five identical rectangular parallel mini-channels have been machine etched into Lexan ® polycarbonate top. They are mated with an aluminum bottom plate. An Iconel-625 strip is placed beneath the aluminum plate and has the same dimensions of 6.35 mm wide and 30.72 cm long. An additional Teflon ® strip of identical dimension is placed under the heating element to assure good thermal contact with the aluminum plate and provide electrical insulation. A Sorensen DC S8-125E power supply with voltage ranging from 0 to 60 volts and current ranging from 0 to 125A is used for resistance heating of the Iconel-625 strip. Each flow channel has a hydraulic diameter of 1.03 mm. Two Omega PX26-015DV pressure transducers are used to measure the overall pressure drop between the inlet and the outlet of the channels.
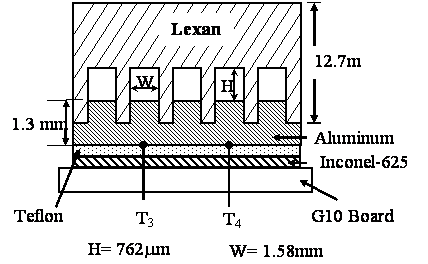
Figure 3. Cross-section of the minichannels
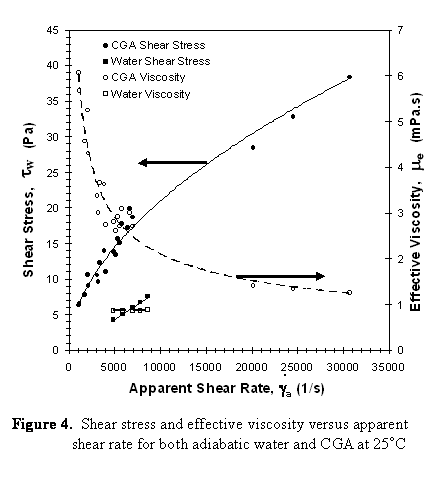
The following conclusions for can be drawn:
- CGA behaves as a shear thinning fluid.
- The friction factor for CGA in laminar flow follows the same law as single phase, i.e., f=15.5/Re with the Reynolds number computed from the effective viscosity of CGA.
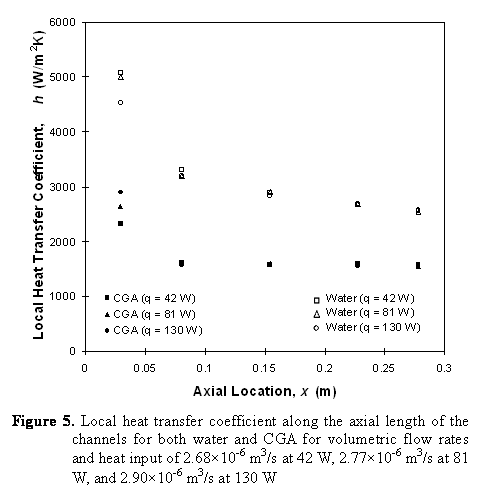
- the local and average heat transfer coefficients for CGA under imposed heat flux and laminar flow conditions are independent of the mass flow rate and heat flux.
- The heat transfer coefficients are smaller than those for water due to the high porosity of the CGA resulting in reduced specific heat and thermal conductivity.
Relevant Publications
H. Tseng, L. Pilon, and G. R. Warrier, 2006. Rheology and Convective Heat Transfer of Colloidal Gas Aphrons in Horizontal Minichannels, International Journal of Heat and Fluid Flow, Vol. 27, No. 2 , pp. 298-310. doi:10.1016/j.ijheatfluidflow.2005.08.009
S. Larmignat, D. Vanderpool, H. Lai, and L. Pilon, 2008. Rheology of Colloidal Gas Aphrons (Microfoams). Colloids and Surfaces A: Physicochemical and Engineering Aspects, Vol.322. pp. 199-210. doi:10.1016/j.colsurfa.2008.03.010
J. Zhao, S. Pillai, and L. Pilon, 2009. Rheology of Microfoams Made From Ionic and Non-Ionic Surfactant Solutions, Colloids and Surfaces A: Physicochemical and Engineering Aspects, Vol. 348, pp. 93–99 . doi: 10.1016/j.colsurfa.2009.06.037